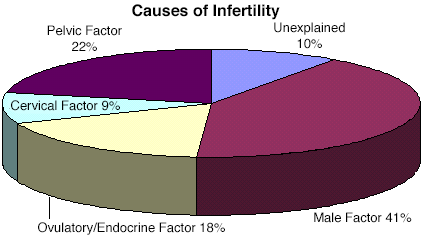
There are a number of fertility tools available to help pinpoint the cause of infertility. After undergoing evaluation through a comprehensive physical exam and medical history, a fertility specialist will initiate a series of specific diagnostic investigations based upon his/her suspicion of the source of infertility. Causes of infertility can generally be classified within the following areas: the female eggs and endocrine system, the female pelvic cavity (tubal damage, endometriosis, uterine problems, fibroids), , or semen quality and the male reproductive tract, or timing and technique of sexual relations. A glossary of medical terms is provided at the end of this page to clarify medical terminology commonly used in fertility treatment.
- Female endocrine system investigation
- Pelvic Factor Investigation in the female pelvic cavity to detect disorders such as tubal disease, endometriosis, uterine problems, fibroids, PCOS, ovulatory disorder, diminished ovarian reserve.
- Male Factor Investigation to detect male infertility causes such as low sperm count, decreased sperm motility, abnormal sperm morphology, or male reproductive system disease that reduce male fertility.
- Cervical Factor Investigation and timing and technique of sexual relations.
- Female Endocrine System Investigation
If a fertility specialist suspects that the source of infertility may be related to ovulatory irregularity or other problems within the female endocrine system, the testing protocols outlined below will most likely be undertaken. Treatments for diagnoses within this category are expanded upon further in our Hormonal Therapy section.- Basal Body Temperature Charting (BBT)
This is the simplest and cheapest test for ovulatory evaluation. Elevated progesterone levels during the second half of the menstrual cycle cause the temperature of the body to rise 0.5-1.0 degree Fahrenheit. A BBT chart which demonstrates a 12 to 14 day elevation in temperature after day 11-16 is considered to be normal. Approximately 90% of women can be expected to have ovulated two days before or after the lowest temperature recorded before a sustained rise. If a BBT does not demonstrate a notable shift in temperature phases, it is called monophasic. There is a 20% chance of ovulation occurring despite a monophasic BBT. Likewise, a biphasic recording is generally a healthy indicator, but it does not always mean that normal ovulation has occurred. Luteinized unruptured follicle syndrome (LUFS) and retained ovum syndrome are theorized ovulatory abnormalities thought to occur despite normal hormonal events. Today, temperature and other body measurements are often provided by digital applications that are reasonably, but not always, accurate in tracking ovulation - Endometrial Biopsy
An endometrial biopsy in the mid to late luteal phase (postovulatory day 7 to 12) was a standard test used for several decades. However, it has been shown not to be helpful in diagnosis, and so it is almost never indicated for fertility alone. It is sometimes used to diagnose endometritis or inflammation of the endometrium, or to obtain tissue for other tests. Much more commonly today, a simple blood test for serum progesterone level can be drawn to confirm ovulation. - Urinary Luteinizing Hormone Detection
Urinary Luteinizing Hormone (uLH) detection kits are very helpful for timing diagnostic procedures, inseminations and intercourse. Different tests have different abilities to predict the LH surge. The most sensitive use of the test requires a woman to empty her bladder in the morning, restrict fluids, and then test between 10 AM and 12 noon. The urinary LH surge usually occurs about one to two days prior to the rise in BBT and 12 to 60 hours before ovulation. A shorter range is 22 to 44 hours, with a mean of 30 hours. However, while useful in timing medical interventions, use of uLH testing to time intercourse is not better than regular intercourse 2-3 times per week without any testing. - Ultrasonography
Ultrasonography utilizes sound waves to image the internal status of the uterus, ovaries, ovarian follicles and other pelvic structures. It can be performed either transabdominally or, preferably, transvaginally, and is a very useful clinical tool in evaluating follicular development and ovulation. Different ultrasound probes and machines give somewhat different measurements, but in general follicles will mature and rupture between 17 to 22 mm in size. The loss of follicular size, the loss of clear follicles, and the appearance of fluid in the cul-de-sac are all suggestive of ovulation. The presence of multiple small follicles is indicative of polycystic ovarian disease. Additionally, endometrium in the midluteal phase greater than 8 mm in thickness is usually reflective of normally developed post ovulatory endometrium. - Follicle Stimulating Hormone (FSH) and Estradiol (E2), Antimullerian Hormone (SMH) and Antral Follicle Count (AFC)
The confirmation of absent or abnormal ovulation, when suggested by screening tests, may require additional tests to rule out other endocrinologic diseases or conditions. The most important test is a cycle day 2-4 serum follicle stimulating hormone (FSH) level. Cycle day 1 is the first day the menstrual period begins. The normal upper range for this test is generally 10-13 mIU/mL. Levels below this range are normal, while levels approaching 20 mIU/mL are associated with markedly decreased pregnancy rates. Only an exceedingly small fraction of patients ever get pregnant if their serum FSH level is above 20 mIU/mL. Serum estradiol (E2) is almost always tested at the same time as the FSH level to prevent an inappropriate interpretation of the test results. Serum E2 on cycle day 2-4 should be less than 85 pg/mL. Several studies have demonstrated that even one elevated cycle day 2-4 FSH level is associated with a poorer prognosis, regardless of the results of additional tests. A more recent and better test to screen for ovarian reserve is the Anti Mullerien Hormone (AMH) test. This involves obtaining a serum AMH level on any day in the menstrual cycle although it is often performed early in the cycle. Results are considered abnormal if the level is less than 1.2ng/ml. A third test of ovarian reserve is antral follicle count (AFC). Transvaginal ultrasounds is performed around cycle day 2 to 4 and follicles between 2-10mm in size are counted in both ovaries. A normal result is 8 or more follicles. While none of these tests predict pregnancy rates directly and and do not have fixed levels for normal or abnormal, they can be used to predict response to ovarian stimulation and therefore can have a relationship to eventual pregnancy rates. Both ovarian function and clinical outcome with in vitro fertilization (IVF) can be predicted with statistically significant differences between normal and abnormal responders. - Laparoscopy
Laparoscopy may be performed for diagnostic purposes to identify ovarian follicles and surface irregularities suggesting normal ovulation. The finding of a follicular cyst on the ovary or corpus luteum is suggestive of ovulation. The presence of multiple small follicles is usually diagnostic for polycystic ovarian disease. Laparoscopy is never performed only for this purpose. - Other Female Endocrinologic Tests
Tests for thyroid disease include thyroid stimulating hormone (TSH) alone and, when abnormal, thyroxine (T4). Additional tests and treatment may be performed as indicated. Serum prolactin (PRL) should be obtained if ovulation dysfunction or galactorrhea (breast secretion) are identified. Serum LH may be helpful in diagnosing polycystic ovarian disease. If abnormal androgen levels are suspected, testing assays may include testosterone and dehydroepiandrosterone sulfate (DHAS) as screening tests, and free testosterone (free T4), androstenedione (A), and 17-hydroxyprogesterone (17 OHP) if indicated, and rarely cortisol and adrenocorticotrophic hormone (ACTH). Karyotype may be indicated for individuals with suspected chromosomal abnormalities. - Genetic Tests
Genetic tests of either or both partners may be indicated for individuals with suspected chromosonal or genetic abnormalities or by those in interested in knowing more about potential genetic health or problems.
- Basal Body Temperature Charting (BBT)
- Pelvic Factor Investigation
If your fertility physician suspects a physical or anatomic problem within the women’s pelvis, one or more of the following diagnostic tests are likely to be used:- Ultrasonography and Sonohysterography
Ultrasonography is a common tool utilized in the diagnosis of infertile patients. Ultrasonography is useful for the diagnosis of myomas, adenomyosis, adnexal or pelvic masses, intrauterine polyps, congenital abnormalities, and polycystic ovarian disease. Enlargement of blocked fallopian tubes (hydrosalpinges) can often also be diagnosed by ultrasonography. The recent widespread utilization of sonohysterography has further improved ultrasonography’s role in diagnosis of infertile conditions. Studies have shown that these tests are generally quite accurate. Since this technique is also a simple office procedure, it is often used instead of or in conjunction with hysteroscopy. - Hysterosalpingogram
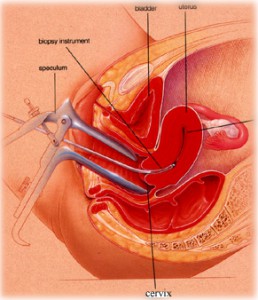
Hysterosalpingogram is a procedure in which a small amount of radio-opaque fluid is injected into the uterus and fallopian tubes and then visualized with x-rays to examine these structures. It has been the primary screening test of pelvic status because it is relatively safe, simple, inexpensive, and reliable. It is useful for diagnosing intrauterine structure and lesions and can evaluate tubal status in the proximal, distal, and sometimes intrapelvic region. Additionally, reasonable data suggest that the HSG may be associated with slightly increased pregnancy rates following its use. Disadvantages include some pain, discomfort, and radiation exposure. Potential complications are rate, but include infection, embolism of dye and hypersensitivity to iodine. - Hysteroscopy
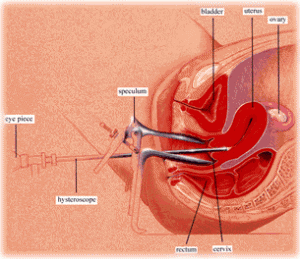 Hysteroscopy is an operative procedure that is performed as a diagnostic procedure or as a therapeutic intervention. The procedure can be done in the office or the operating room, depending upon the method of anesthesia used and the extent of the procedure. Hysteroscopy has the advantage of being a very versatile procedure which avoids ionizing radiation and has a lower false-positive rate than HSG. It can also be used to perform selective hydrotubation of each fallopian tube, tubal catheterization to open blocked tubes, and rarely, falloposcopy to look inside fallopian tubes. These diagnostic tests may be very helpful in determining the functional status of the fallopian tubes. Disadvantages to hysteroscopy is the fact that it is an operative procedure with attendant potential surgical and anesthetic complications. This procedure can usually be done with a gentle intravenous anaesthesia known as conscious sedation.
Hysteroscopy is an operative procedure that is performed as a diagnostic procedure or as a therapeutic intervention. The procedure can be done in the office or the operating room, depending upon the method of anesthesia used and the extent of the procedure. Hysteroscopy has the advantage of being a very versatile procedure which avoids ionizing radiation and has a lower false-positive rate than HSG. It can also be used to perform selective hydrotubation of each fallopian tube, tubal catheterization to open blocked tubes, and rarely, falloposcopy to look inside fallopian tubes. These diagnostic tests may be very helpful in determining the functional status of the fallopian tubes. Disadvantages to hysteroscopy is the fact that it is an operative procedure with attendant potential surgical and anesthetic complications. This procedure can usually be done with a gentle intravenous anaesthesia known as conscious sedation. - Magnetic Resonance Imaging
Magnetic resonance imaging is rarely needed for identification of pelvic organs or lesions. However, it can be useful for differentiating adenomyosis from myomas, and for complex congenital uterine abnormalities as well as other rarer uterine abnormalities or pelvic masses. Its high cost limits its general use, but it is helpful in selected situations. - Laparoscopy and Hydrotubation
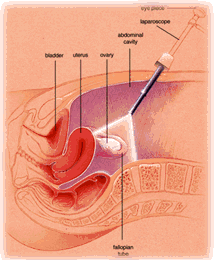
Diagnostic laparoscopy can be performed with small instruments under conscious sedation in a properly equipped procedure room or in an operating room under general anesthesia. The procedure provides excellent diagnostic capabilities to evaluate the uterus, fallopian tubes, ovaries and pelvic surfaces. The primary diagnoses are tubal occlusion, adhesions, endometriosis, fibroids, congenital anomalies, and polycystic ovarian disease. Other intraabdominal organs can also be evaluated. Laparoscopy under conscious sedation provides very limited opportunities for therapeutic intervention if abnormalities are detected. The major advantages of laparoscopy is its accuracy in diagnosing infertility conditions and its therapeutic potential. The major disadvantage is cost and the fact it is an invasive procedure with the attendant potential complications. Laparoscopy may be performed following an abnormal hysterosalpingogram, sonohysterography, or hysteroscopy if the information acquired at laparoscopy will likely alter the treatment plan, or if it is expected that a condition can be effectively treated during the procedure.
- Ultrasonography and Sonohysterography
- Male Factor Investigation
If your fertility physician suspects that the source of infertility may be related to poor semen quality or other issues with the male reproductive tract, (s)he will most likely make use of the following tests:
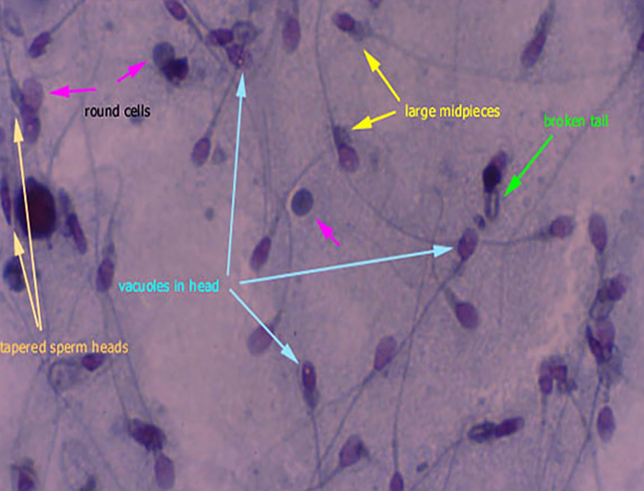
- Semen Analysis
Semen analysis is the most direct, simple and cost-effective test of male fertility. At least two tests should be obtained four weeks apart unless the male has sired (impregnated a woman) within the past three years, in which case one test is sufficient if it is normal. Semen volume should be at least 1.5mL. Sperm concentration at least 15 million/mL, 39 million in total ejaculate, motility at least 32% progressively motile at 2 hours and, 40% total motility and 4.0% morphologically normal by strict criteria. Borderline test results need to be interpreted cautiously since there are few data supporting the use of absolute cut-off points in borderline ranges. Extremely poor test results call for further intervention. - Sperm Fragmentation Assay
Sperm DNA damage is more common in infertile men and may contribute to poor reproductive performance. Sperm DNA damage is also associated with spontaneous recurrent miscarriage. Because the prognostic clinical value of DNA integrity testing may not affect treatment, the routine use of DNA tests is not recommended. - Additional Tests
For men with azoospermia, seminal plasma fructose should be obtained to document the status of the vas deferens. For men with azoospermic or severely abnormal parameters, FSH, testosterone and prolactin should be measured and testicular biopsy should be considered. Hormonal evaluation should also be performed in the presence of male sexual dysfunction. Transcrotal and scrotal ultrasound are indicated in some cases. Karyotype and other genetic tests may be indicated for individuals with suspected chromosomal abnormalities. All men with very poor semen analysis, e.g. sperm count less than 10 million per mL, should be referred to a urologist. - Cervical Factor Investigation and Timing and Technique of Sexual Relations
If your fertility specialist suspects that the source of infertility may be related to the interactions of the cervical mucus with the sperm or the timing and technique of sexual relations, (s)he will most likely make use of the testing protocols outlined below.- Post-Coital Test
The post-coital test was the standard test for evaluating cervical factor infertility for decades. However, it has been shown not to be predictive of pregnancy rate and is no longer used except in highly specific clinical situations. - Female Cervical and Male Urethral Cultures
Female cervical and male urethral cultures for E. coli, gonorrhea, chlamydia, and mycoplasma can be helpful in identifying infection in selected patients or in those undergoing intrauterine insemination or assisted reproductive technology procedures.These infertility screening tests are mostly of historical interest and are very rarely used.
- Post-Coital Test
- Semen Analysis
